Boisar, Mumbai, Maharashtra
- GST NO. : 27AAKCM1451A1ZV
All You Need To Know About Isothiocyanate
Posted by Admin on September, 12, 2020
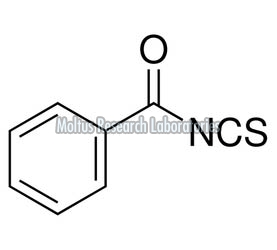
As per isothiocyanate & thiochloroformate manufacturers, Isothiocyanate is a special chemical which is formed replacing the oxygen atom present in the isocyanate with sulphur. There are several types of isothiocyanates which are found from various natural sources like planta. Some plants produce an enzyme called as metabolites which are converted systematically to form glucosinolates. The most common type of naturally occurring isothiocyanates is known as allyl isothiocyanate, more popularly known as the mustard oil.
Another type of isothiocyanate produced using advanced methods is known as phenyl isothiocyanate. It is used for undertaking the process of sequencing amino acid during 'Edman degradation' procedure.
Formation of phenyl isothiocyanate:
The method which is followed for the formation of the isothiocyanates starts with a series of reaction between the primary amine like aniline with carbon disulfide. The reaction occurs between these two compounds in an aqueous solution of ammonia. The reaction leads to the formation of precipitates which is actually a special salt known as ammonium dithiocarbamate.
This precipitate or salt is treated with the lead nitrate solution to result in the formation of isothiocyanate. Apart from this method, one more process is followed to make isothiocyanate. In this process, tosyl chloride is used for the decomposition process of the dithiocarbamate salts which is formed in the first case.
Synthesis of the formed Phenyl Isothiocyanate:
Coming to the synthesis of the phenyl isothiocyanates, it can be easily accessed using a special reaction procedure which is known as thermally induced fragmentation reaction. The reaction takes place between different oxathiazoles. This is a special method of treating this polymer compound, isothiocyanates. It is basically a weak electrophile which is akin to the reactions of carbon dioxide whereas nucleophiles do attack the carbon elements.
The reaction between the acetophenone enolate and phenyl isothiocyanate is commonly known as one-pot synthesis. It is regarded as the ultimate reaction in which the product called Thiazolidine is formed. This particular reaction is considered very stereoselectivedue to the formation of the isomer Z. As the isothiocyanates are slightly electrophilic in character, they are quite susceptible to the process of hydrolysis.
Presence of isothiocyanate in nature
Isothiocyanateis found widely in nature and has become quite popular in the field of science and medication related to food products. Vegetable foods gain certain kinds of special flavors due to the presence of isothiocyanates. The food products which contain these compound includes wasabi, radish, horseradish, Brussels sprouts, mustard, capers, watercress, nasturtiums, and papaya seeds. The plants of these vegetable foods are those species which generate isothiocyanates in different amounts.
As they produce similar kind of isothiocyanate, although different in being, they have a similar kind of flavor. For example, all the members belonging to the order of Brassicales are classified and characterized on the basis of how glucosinolatesis produced. Myrosinase, which is an enzyme, is produced and it acts on glucosinolates which result in the formation or release of Isothiocyanate & thiochloroformate.
Search
Category
Recent Posts
- High Purity and Reliable Industrial Supply of Benzyl Isothiocyanate & Benzoyl Isothiocyanate Manufacturers
- Global 4-Isothiocyanato-2-(trifluoromethyl) Benzonitrile Manufacturers
- A Complete Guide to Leading 4-Isothiocyanato-2-(trifluoromethyl) Benzonitrile Manufacturers for Industrial Applications
- Why You Should Buy 4-Isothiocyanato-2-(trifluoromethyl) Benzonitrile from Reputed Manufacturers Only
- 1,1′-Thiocarbonyldiimidazole – Applications, Properties, and Industrial Importance


Leave a Comment