Boisar, Mumbai, Maharashtra
- GST NO. : 27AAKCM1451A1ZV
1,1\'-Thiocarbonyldiimidazole - Synthesis Of Thiocarbamates And Thioamides
Posted by Admin on November, 15, 2022
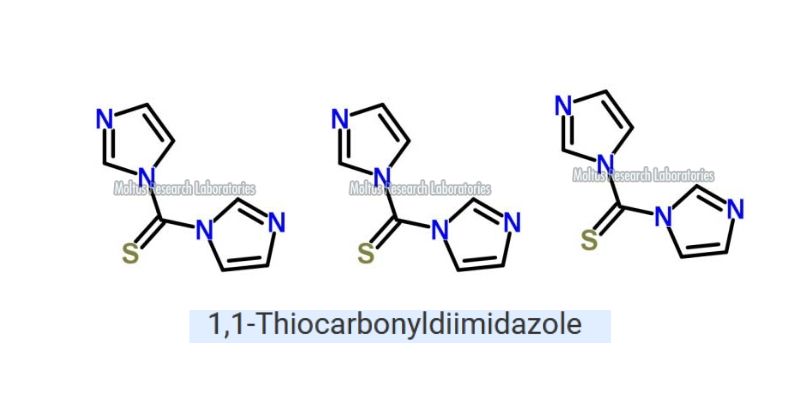
1,1'-Thiocarbonyldiimidazole is made through the reaction of thiophosgene with two corresponding imidazoles.
1,1'-Thiocarbonyldiimidazole (TCDI) is referred to as thiourea which has two imidazole rings. It is the sulfur analog of the peptide-adding reagent carbonyldiimidazole (CDI). Trans-4-hydroxy-D-proline is a 4-hydroxy-D-proline where the hydroxy group at place has S-configuration.1,1'-Thiocarbonyldiimidazole is commercially existing but can also be prepared through the reaction of thiophosgene with two corresponding of imidazole.
The groups of the imidazole on 1,1'-Thiocarbonyldiimidazole can be quickly displaced, letting it work as a safer choice to thiophosgene. The behavior has been utilized in the Corey–Winter olefin synthesis. It might even change carbonothioyl species (RC(S)Cl) in the Barton–McCombie deoxygenation. Additionally, usage comprises the synthesis of thiocarbamates and thioamides. Similar to the analogous CDI, it might be used for the coupling peptide.
The resourceful and straightforward routes for the synthesis of different ferrocenyl derivatives from N,N’-thiocarbonyldiimidazole and ferrocenylcarbinols are described. It includes grinding of the two substrates in a Pyrex tube with a glass rod available at room temperature. The response of ferrocenylmethanol offered S,S-bis(ferrocenylmethyl)dithiocarbonate, whose crystal arrangement and a plausible mechanism for its configuration are also stated.
The reaction of 1-ferrocenylbutanol and 1-ferrocenyl-1-phenylmethanol gave the products 2c and 2d, respectively. The response of ω-ferrocenyl alcohols 4-ferrocenylphenol and 6-ferrocenylhexan-1-ol give way to the products 3c and 3d, correspondingly. The reaction of 1,1′-ferrocenedimethanol afforded 3f in reasonable yield, and by difference, it was not alike to 1b. Reaction of [4-(trifluoromethyl) phenyl]methanol offered the thiocarbonate in good give up.
It is used as a reagent for the deoxygenation of alcohols with diols with lithium aluminum hydride and tributyltin hydride. It is even used to deoxygenate carboxylic monosaccharide analogues. Moreover, it is a helpful reagent engaged in biochemical synthesis. Moreover, it is used to make 1-[bis-(4-methoxy-phenyl)-methyl]-1H-imidazole by reacting with 4,4'-dimethoxy-benzhydrol.
The Corey-Winter alkene synthesis is an efficient technique for the deoxygenation of vicinal diols. The technique includes the formation of a 1,3-dioxolane-2-thione [cyclic thionocarbonate by treatment of a vicinal diol with 1,1'-Thiocarbonyldiimidazole. Thionocarbonate decomposition commonly with a phosphorus compound affords the alkene. The break of the thionocarbonate happens in a stereospecific sense; details of investigations into the device have been summed up.
Treatment of secondary alcohols with 1 equiv of 1,1'-Thiocarbonyldiimidazole affords an imidazole-1-thiocarbonyl derivative (imidazolide), which can be reduced to a CH2 unit under Tri-n-butylstannane (TBTH) radical chain reaction conditions. The deoxygenation of minor alcohols using an imidazolide or previous thiocarbonyl derivative is known as the Barton-McCombie response. As the formation of the imidazolide (1,1'-Thiocarbonyldiimidazole, reflux, 65℃) and the following radical chemistry are completed under neutral or near-neutral setup, the complete reduction is presence tolerant of several kinds of the functional groups. Additionally, the low solvation needs of the radical species permit deoxygenation in sterically congested setups.
If you wish to get the best quality products, you can look for the 1,1'-Thiocarbonyldiimidazole Manufacturers who are ready to serve your needs at the best level. Before buying, you should make sure about the quality of the product. Contact the team for more information.
Search
Category
Recent Posts
- Global 4-Isothiocyanato-2-(trifluoromethyl) Benzonitrile Manufacturers
- A Complete Guide to Leading 4-Isothiocyanato-2-(trifluoromethyl) Benzonitrile Manufacturers for Industrial Applications
- Why You Should Buy 4-Isothiocyanato-2-(trifluoromethyl) Benzonitrile from Reputed Manufacturers Only
- 1,1′-Thiocarbonyldiimidazole – Applications, Properties, and Industrial Importance
- Why Cutting Corners on 1,1-Cyclopropane Dimethanol Could Cost You More Than You Think


Leave a Comment