Boisar, Mumbai, Maharashtra
- GST NO. : 27AAKCM1451A1ZV
Thiophosgene: - An Overview
Posted by Admin on January, 24, 2020
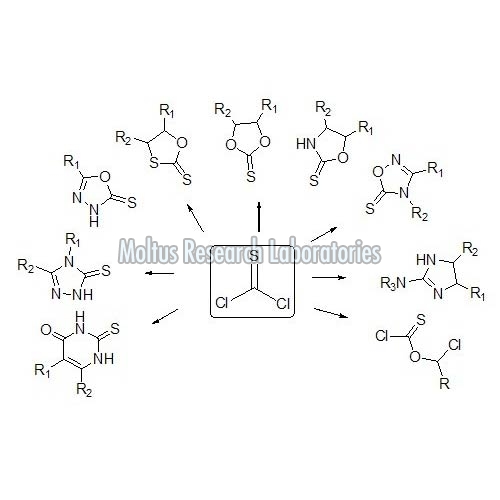
Thiophosgene is a very short-lived compound found in the form of red liquid. It is a molecule with trigonal planar geometry and has a broad spectrum of reaction with a variety of functional groups. The chemical formula of thiophosgene is CSCl2 and the two reactive C-Cl bonds that allow the compound to be used in diverse organic syntheses. It is a reagent used to prepare a bulky thiourea ligand for palladium-catalyzed aerobic oxidation of alcohols to aldehydes and ketones. The compound is volatile and reacts with water to form carbonyl sulfide (COS) and two molecules of hydrogen chloride. The formed carbonyl sulfide then reacts with another water molecule to form hydrogen sulfide and carbon dioxide. Basically, thiophosgene has two reactive sites associated with the carbon atom. Both chlorine atoms of thiophosgene are electronegative, the carbon atom becomes positively charged, thus creating an electrophile. The compound prepared by reductive dechlorination of trichloromethanesulfenyl chloride.
Alternate chemical names of Thiophosgene:-
• CARBONIC DICHLORIDE
• CARBON CHLOROSULFIDE
• CARBONYL SULFIDE DICHLORIDE
• DICHLOROTHIOCARBONYL
• DICHLOROTHIOFORMALDEHYDE
• THIOCARBONIC DICHLORIDE
• THIOCARBONYL CHLORIDE
• THIOCARBONYL DICHLORIDE
How Thiophosgene is formulated:-
Thiophosgene (CSCl2) is prepared in a two-step process from carbon disulfide. The carbon disulfide is chlorinated in the first step to give trichloromethanesulfenyl chloride (perchloromethyl mercaptan), CCl3SCl:
CS2 + 3 Cl2 → CCl3SCl + S2Cl2
The chlorination is controlled as excess chlorine converts trichloromethanesulfenyl chloride into carbon tetrachloride. The trichloromethanesulfenyl chloride is separated by steam distillation. Trichloromethanesulfenyl chloride is a rare sulfenyl chloride, and the process hydrolyzes the sulfur monochloride. Reduction of trichloromethanesulfenyl chloride gives thiophosgene:
CCl3SCl + M → CSCl2 + MCl2
Properties of thiophosgene:-
• Molar mass of thiophosgene is 114.98g/mol.
• Usually, the chemical found in the form of red liquid.
• The density of the compound is 1.50 g/cm3.
• The boiling point of the chemical is 70 to 75 0C.
• It decomposes and has low water solubility.
Uses of Thiophosgene:-
• Thiophosgene is used to prepare chemicals like isothiocyanates, thioureas, thiocarbamates, and thiocarbonates. It is also used to produce tolnaftate (Antifungal Drug).
• It is extensively used to synthesize a huge variety of thioxo substituted heterocycles of pharmaceutical importance by cyclisation of phenols, amines, alcohols, and thiols.
• The compound is also used to prepare many inorganic complexes with the conversion of phenols, thioureas, amides, urea and thioamides to thiophenols, respectively.
• Thiophosgene is reacted with dienes and alkenes to formulate bridged compounds and different reactive synthons.
Disadvantages of Thiophosgene:-
• Thiophosgene reacts with water to produce hydrogen chloride, carbon disulfide, and carbon dioxide and the reaction is slow unless the water is hot.
• Toxic phosgene, sulfur dioxide and hydrogen chloride produced from thiophosgene may be generated in a fire.
• Inhalation of this chemical cause irritation of the respiratory system and delayed pulmonary edema.
• The vapor from thiophosgene irritates eyes.
• The chemical burns skin and eyes.
• Ingestion of the chemical cause irritation of mouth and stomach.
• The chemical is incompatible with acids, diazo and azo compounds, aldehydes, alkali metals, halocarbons, isocyanates, nitrides, hydrides, and other strong reducing agents. The reactions of thiophosgene with these mentioned compounds generate heat and in many cases hydrogen gas. The generated hydrogen sulfide often reacts with acids.
A wide range of this chemical is presented by thiophosgene manufacturers in India. With the core expertise founded in synthetic and process chemistry, the leading manufacturers produce thiophosgene to meet the requirements of the clients in the market.
Search
Category
Recent Posts
- Global 4-Isothiocyanato-2-(trifluoromethyl) Benzonitrile Manufacturers
- A Complete Guide to Leading 4-Isothiocyanato-2-(trifluoromethyl) Benzonitrile Manufacturers for Industrial Applications
- Why You Should Buy 4-Isothiocyanato-2-(trifluoromethyl) Benzonitrile from Reputed Manufacturers Only
- 1,1′-Thiocarbonyldiimidazole – Applications, Properties, and Industrial Importance
- Why Cutting Corners on 1,1-Cyclopropane Dimethanol Could Cost You More Than You Think


Leave a Comment